 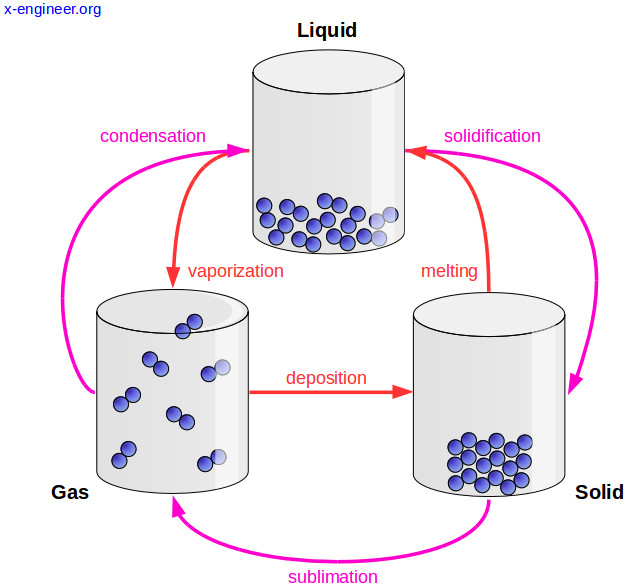
The average amount of empty space between molecules gets progressively larger as a sample of matter moves from the solid to the liquid and gas phases. There are spaces between particles of matter. A change in phase may occur when the energy of the particles is changed. The temperature of a substance is a measure of the average kinetic energy of the particles. 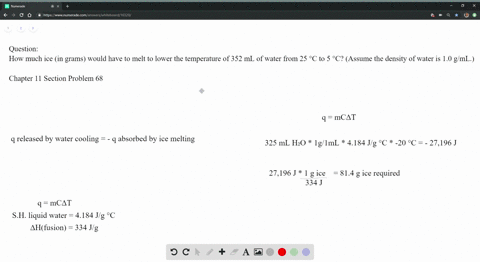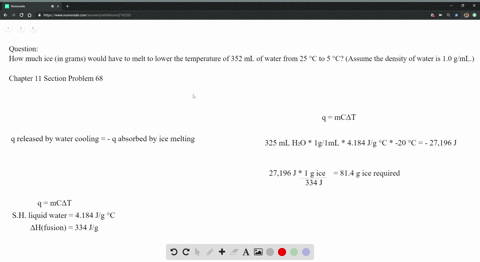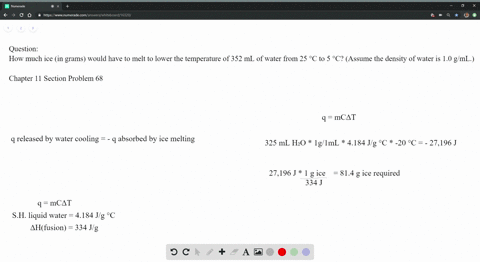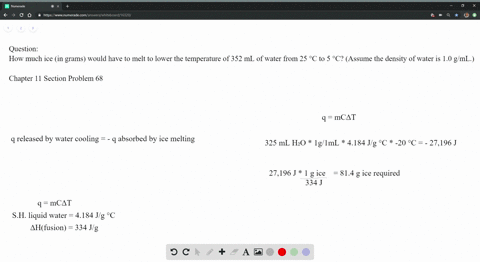 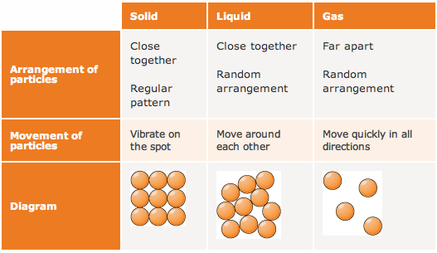
Molecules in the solid phase have the least amount of energy, while gas particles have the greatest amount of energy. This in turn determines whether the substance exists in the solid, liquid, or gaseous state. All particles have energy, but the energy varies depending on the temperature the sample of matter is in. 
Matter is made up of particles that are constantly moving. The kinetic molecular theory of matter states that: Kinetic Molecular Theory The kinetic molecular theory of matter explains how matter can change among the phases of solid, liquid, and gas. In this chapter, the nature of these interactions and physical properties of liquid and solid phases will be examined. In the liquid and solid states, these interactions are of considerable strength and play an important role in determining a number of physical properties that do depend on the chemical identity of the substance. The changes at high pressures and low temperatures- conditions that permit the atoms and molecules to interact to a much greater extent. The great distances between atoms and molecules in a gaseous phase, and the corresponding absence of any significant interactions between them, allows for simple descriptions of many physical properties that are the same for all gases, regardless of their chemical identities. Describe and differentiate the types of intermolecular forces. Use the kinetic molecular model to explain properties of liquids and solids. London Dispersion Forces Dipole-Dipole Forces Ion-Dipole Forces Hydrogen Bonding Have the particles held by Intermolecular Forces CHAPTER 1 KINETIC MOLECULAR MODEL OF LIQUIDS AND SOLIDS & INTERMOLECULAR FORCESĬONCEPT MAP Kinetic Molecular Model Explains the properties of 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
